Nori Chicken IL-4 ELISA Kit
Price range: $508.00 through $916.00
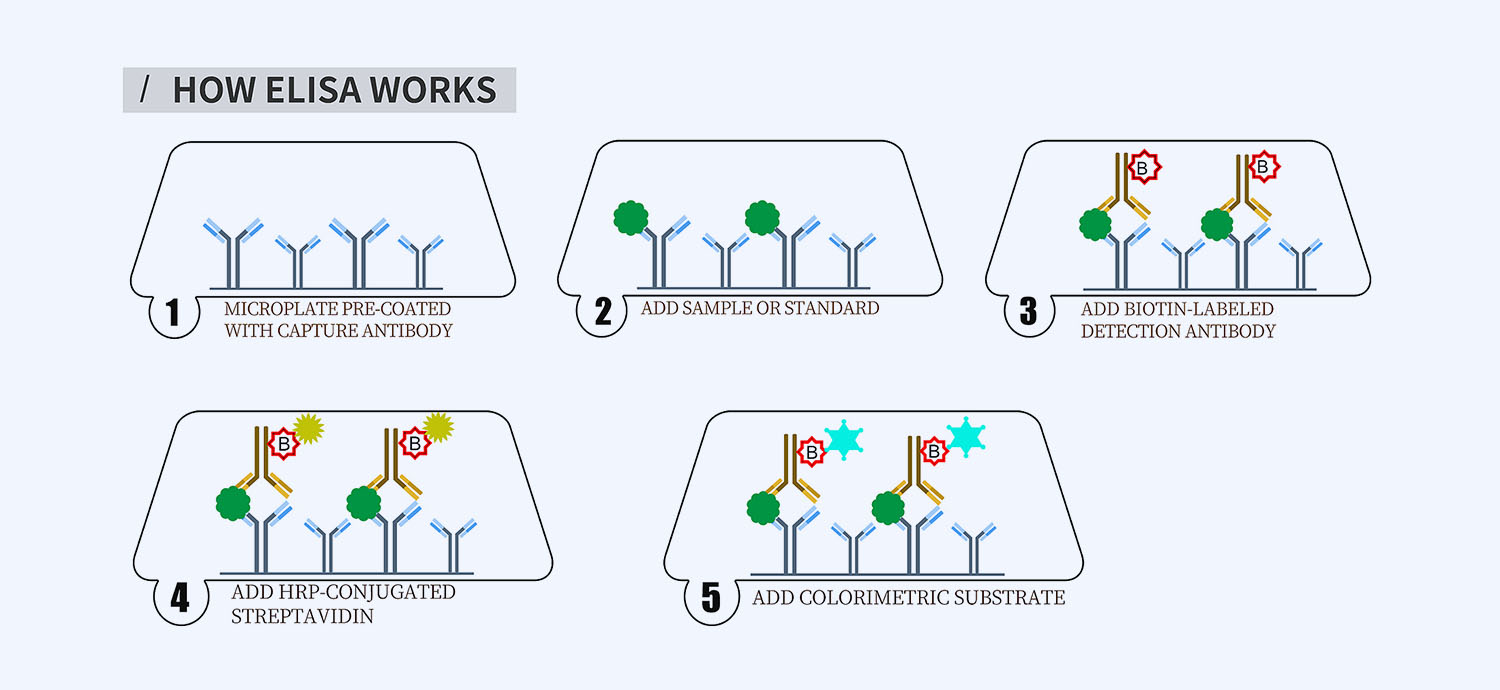
This ELISA kit is for quantification of IL-4 in chicken. This is a quick ELISA assay that reduces time to 50% compared to the conventional method, and the entire assay only takes 3 hours. This assay employs the quantitative sandwich enzyme immunoassay technique and uses biotin-streptavidin chemistry to improve the performance of the assays. An antibody specific for IL-4 has been pre-coated onto a microplate. Standards and samples are pipetted into the wells and any IL-4 present is bound by the immobilized antibody. After washing away any unbound substances, a detection antibody specific for IL-4 is added to the wells. Following wash to remove any unbound antibody reagent, a detection reagent is added. After intensive wash a substrate solution is added to the wells and color develops in proportion to the amount of IL-4 bound in the initial step. The color development is stopped, and the intensity of the color is measured.
Alternative names for IL-4: Interleukin 4, IL4
This product is for Laboratory Research Use Only not for diagnostic and therapeutic purposes or any other purposes.
- Description
- How Elisa Works
- Product Citations (18)
- Reviews (0)
Description
Nori Chicken IL-4 ELISA Kit Summary
Alternative names for IL-4: Interleukin 4, IL4
| Assay Type | Solid Phase Sandwich ELISA |
| Format | 96-well Microplate or 96-Well Strip Microplate |
| Method of Detection | Colorimetric |
| Number of Targets Detected | 1 |
| Target Antigen Accession Number | C4PAA9 |
| Assay Length | 3 hours |
| Quantitative/Semiquantitative | Quantitative |
| Sample Type | Plasma, Serum, Cell Culture, Urine, Cell/Tissue Lysates, Synovial Fluid, BAL, |
| Recommended Sample Dilution (Plasma/Serum) | No dilution for sample <ULOQ; sufficient dilution for samples >ULOQ |
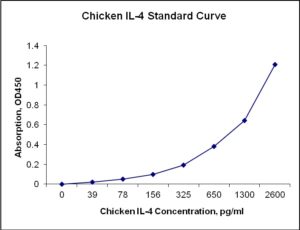
| Sensitivity | 8 pg/mL |
| Detection Range | 39-2600 pg/mL |
| Specificity | Chicken IL-4 |
| Cross-Reactivity | < 0.5% cross-reactivity observed with available related molecules, < 50% cross-species reactivity observed with species tested. |
| Interference | No significant interference observed with available related molecules |
| Storage/Stability | 4 ºC for up to 6 months |
| Usage | For Laboratory Research Use Only. Not for diagnostic or therapeutic use. |
| Additional Notes | The kit allows for use in multiple experiments. |
Standard Curve
Kit Components
1. Pre-coated 96-well Microplate
2. Biotinylated Detection Antibody
3. Streptavidin-HRP Conjugate
4. Lyophilized Standards
5. TMB One-Step Substrate
6. Stop Solution
7. 20 x PBS
8. Assay Buffer
Other Materials Required but not Provided:
1. Microplate Reader capable of measuring absorption at 450 nm
2. Log-log graph paper or computer and software for ELISA data analysis
3. Precision pipettes (1-1000 µl)
4. Multi-channel pipettes (300 µl)
5. Distilled or deionized water
Protocol Outline
1. Prepare all reagents, samples and standards as instructed in the datasheet.
2. Add 100 µl of Standard or samples to each well and incubate 1 h at RT.
3. Add 100 µl of Working Detection Antibody to each well and incubate 1 h at RT.
4. Add 100 µl of Working Streptavidin-HRP to each well and incubate 20 min at RT.
5. Add 100 µl of Substrate to each well and incubate 5-30 min at RT.
6. Add 50 µl of Stop Solution to each well and read at 450 nm immediately.
Background:
Interleukin-4 (IL-4) is a cytokine that is expressed in a variety of tissues including lymphocytes and leukocytes, is a key regulator in humoral and adaptive immunity and has many biological roles, including the stimulation of activated B-cell and T-cell proliferation, and the differentiation of CD4+ T-cells into Th2 cells. IL-4 induces differentiation of naive helper T cells (Th0 cells) to Th2 cells. Upon activation by IL-4, Th2 cells subsequently produce additional IL-4. The cell that initially produces IL-4, thus inducing Th0 differentiation, has not been identified, but recent studies suggest that basophils may be the effector cell.[1] It is closely related and has functions similar to Interleukin 13. Like IL-13, Interleukin 4 (IL-4) is critical for responses to parasitic helminthes. [2] IL-4 up-regulates MHC class II production and decreases the production of Th1 cells, macrophages, IFN-gamma, and dendritic cell IL-12. Tissue macrophages play an important role in chronic inflammation and wound repair. The presence of IL-4 in extravascular tissues promotes alternative activation of macrophages into M2 cells and inhibits classical activation of macrophages into M1 cells. An increase in repair macrophages (M2) is coupled with secretion of IL-10 and TGF-β that result in a diminution of pathological inflammation. This cytokine was co-discovered by Maureen Howard and William Paul[3] and by Dr. Ellen Vitetta and her research group in 1982. The nucleotide sequence for human IL-4 was isolated four years later confirming its similarity to a mouse protein called B-cell stimulatory factor-1 (BCSF-1).[4]
References
- Sokol, C.L., et al. (2008). Nat Immunol 9 (3): 310–318.
- Liang, H-E, et al. (2012) Nature Immunology, 13: 58–66.
- Howard M, Paul WE (1982). “Interleukins for B lymphocytes”. Lymphokine Res. 1 (1): 1–4.
- Yokota T et al. (1986). Proc. Natl. Acad. Sci. U.S.A. 83 (16): 5894–8.
Product Citations
- Da Silva et al. (2014) Horse seropositive for neospora spp.: Immunoglobulin, cytokines, and c-reactive protein levels. J Equine Vet Sci. 34(10):1240-143. Impact factor: 0.882. Products used and cited: Nori Equine TNF-α, IFNg, IL-1β, IL-1RA, IL-4, IL-6 and IL-10 ELISA kits.
- Do Carmo GM (2015) Immunological response and markers of cell damage in seropositive horses for Toxoplasma gondii. Comparative Immunology, Microbiology and Infectious Diseases. 38:9-13. Impact factor: 1.875. Products used and cited: Nori Equine IL-1β, IL-4, IL-6, IL-10, IFNg and TNF-α ELISA Kits
- Von Antonella Victoria Block (2015) Studie Zur wiederkehrenden atemwegsobstruktium des pferdes (RAO): potentieller pathophysiologischer stellenwert von vas kularem endothelialem wachstumsfaktor (VEGF), tumornekrosefaktor-alpha (TNF-α) und vasoaktivem intestinalem peptid (VIP). Page 23. Products used and cited: Nori Equine IL-4, IL-6, IL-8, TNF-α and VEGF ELISA kits.
- Canciello A et al. (2017) Progesterone prevents epithelialmesenchymal transition of ovine amniotic epithelial cells and enhances their immunomodulatory properties. Nature, 7:3761. DOI:10.1038/s41598-017-03908-1. Impact factor: 40.137. Products used and cited: Nori Sheep IL-4, IL-10 and TGFb3 ELISA Kits, GR106451, GR106454, GR106138.
- Rajput IR et al. (2017) Saccharomyces boulardii and Bacillus subtilis B10 modulate TLRs and cytokines expression patterns in jejunum and ileum of broilers. PLOS ONE 12(3):e0173917. Impact factor: 2.806. Products used and cited: Nori Chicken IL-12, IL-17, IL-4, IFNg and IL-8 ELISA Kits.
- Wu Y et al. (2018) Effects of dietary enterococcus faecium NCIMB11811 supplementation on growth performance and cellular and humoral immune responses in broiler chickens. Poultry Science http://dx.doi.org/10.3382/ps/pey368. Impact factor: 2.35. Products used and cited: Nori Chicken IL-1b, IL-2, IL-4, IL-6, IL-10, IFNg, TNFa ELISA Kits.
- Pi Y et al. (2019) Rubber seed oil and flaxseed oil supplementation on serum fatty acid profile, oxidative-stability of serum and milk and immune function of dairy cows. Asian-Australian J Animal Sci. 32(9):1363-1372. Impact factor: 0.971. Products used and cited: Nori Bovine IL-2, IL-4, IL-6, IL-10, IFNg, TNFa and PGE2 ELISA Kits.
- Sureshkumar S et al. (2019) Oral administration of lactobacillus reuteri expressing a 3D8 single-chain variable fragment (scFv) enhances chicken growth and conserves immune homeostasis. 3 Biotech 9:282. Impact factor: 1.497. Products used and cited: Nori Chicken IL-1β, IL-4, IL-6, IL-8, IFNγ, IGF1, and TNFα ELISA Kits.
- Greco L et al. (2020) Characterization of endocannabinoid system and interleukin profiles in ovine AEC: cannabinoid receptors type-1 and type-2 as key effectors of proinflammatory response. Cells 9, 1008, doi:10:3390/cells9041008. Impact factor: 36.216. Products used and cited: Nori Sheep IL-4, IL-10 and IL-12 ELISA Kits (GR106451, GR106454, GR106407).
- Canciello A et al. (2020) Progesterone prolongs viability and anti-inflammatory functions of explanted preterm ovine amniotic membrane. Frontiers in Bioengineering and Biotechnology 8 Article 135. frontiersin.org. Impact factor: 3.52. Products used and cited: Nori Sheep IL-4, IL-10 and TGFb1 ELISA Kits (GR106451, GR106454, GR106127).
- Sureshkumar S et al. (2020) Administration of L Salivarius expressing 3D8 scFv as a feed additive improved the growth performance, immune homeostasis, and gut microbiota of chickens. Animal Sc J DOI:10.1111/asj.13399. Impact factor: 1.714. Products and cited: Nori Chicken IL-1 beta, IL-6, IL-8, IL-4, IFN gamma, IGF1 and TNF alpha ELISA Kits (GR106294, GR106334, GR114139, GR106340, GR114082, GR106410, GR114119).
- Sureshkumar S. et al. (2020) Inclusion of Lactobacsllus salivarius strain revealed a positive effect on improving growth performance, fecal microbiota and immunological responses in chicken. Archives of Microbiologyorg/10.1007/s00203-020-02088-3. Impact factor: 1.607. Products used and cited: Nori Chicken IL-4, IL-6, IL-8, IL-1b, TNFa, IFNg and IGF1 ELISA Kits.
- Song B et al. (2021) Effects of age on immune function in broiler chickens. J Animal Sci Biotech 12:42. Impact factor: 4.167. Products used and cited: Nori Chicken IL-1β, IL-2, IL-4, IL-10 and IFNγ ELISA Kits.
- Sureshkumar S et al. (2022) Preliminary Study to Investigate the Effect of Lactobacillus Reuteri Administration on Growth Performance, Immunological, Gut Microbiome and Intestinal Mucosa of Chicken. Brazilian J Poult Sci 24(4):001-006. Impact factor: 0.9. http://dx.doi.org/10.1590/1806-9061-2022-1640. Products used and cited: Nori Chicken TNFa, IL-4, IL-6, IL-8, IL-1B, IFNg, IGF1 ELISA Kit.
- Sureshkumar S et al. (2021) Inclusion of Lactobacillus salivariusstrain revealed a positive effect on improving growth performance, fecal microbiota and immunological responses in chicken. Archives of Microbiology 230:847-853. Impact factor: 2.8. Products used and cited: Nori Chicken IFNg, IL-8, TNFa, IL-4 ELISA Kit.
- Adamic N et al. (2022) Effect of intrabronchial administration of autologous adipose‑derived mesenchymal stem cells on severe equine asthma. Stem Cell Res & Therapy 13:23. https://doi.org/10.1186/s13287-022-02704-7. Impact factor: 7.9. Products used and cited: Nori Equine IL-1B, IL-4, IL-8, IL-17, TNFa, IFNg ELISA Kit.
- Song B et al. (2022) Comparison and Correlation Analysis of Immune Function and Gut Microbiota of Broiler Chickens Raised in Double-Layer Cages and Litter Floor Pens. Microbiol Spectrum 10(4). https://doi.org/10.1128/spectrum.00045-22. Impact factor: 8. Products used and cited: Nori Chicken IL-1B, IL-4, IL-10, IFNg ELISA Kit.
- Song B et al. (2022) Effects of Dietary Astragalus Polysaccharide Supplementation on the Th17/Treg Balance and the Gut Microbiota of Broiler Chickens Challenged With Necrotic Enteritis. frontiers in Immunol https://doi.org/10.3389/fimmu.2022.781934. Impact factor: 6.8. Products used and cited: Nori Chicken IL-1B, IL-4, IL-10, IL-17, IFNg ELISA Kit.
Be the first to review “Nori Chicken IL-4 ELISA Kit”
You must be logged in to post a review.






















Reviews
There are no reviews yet.